Introduction to HIV (Part 2)
Transmission
HIV is not able to enter through most of the surface linings of the body but can be passed to another person through mucous ‘wet’ membranes or damaged tissue or directly into the blood stream. HIV is present in the blood, semen, pre-seminal fluid, vaginal and rectal fluids and breastmilk of infected people. The level of virus in some fluids, such as saliva, is too low to be infectious. There are a number of ways in which a person can become infected with HIV:
Unprotected sexual intercourse with an infected partner – anal, vaginal and oral
Injection or transfusion of contaminated blood or blood products
Sharing injection equipment that has been used by someone infected with HIV
Mother to child transmission – during pregnancy, at birth and through breastfeeding
Occupational exposure of healthcare staff
Having other sexually transmitted infections such as herpes, syphilis and gonorrhoea can increase the risk of transmission. Genital ulcers can cause breaks in the genital tract lining or skin which can be an entry point for HIV and the associated local inflammation increases the number of cells in the secretions that can be targets for the virus.
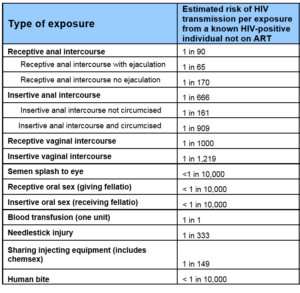
Below is a table from the BASHH (British Association of Sexual Health & HIV) guidelines detailing the risk of developing HIV following exposure from a known HIV positive person 4
HIV testing
HIV antibody production begins around two weeks after transmission. HIV specific antibodies can be detected after four to six weeks in the majority of cases, and all available HIV tests are based on antigen-antibody binding.
Most clinics use newer 4th and 5th generation tests, which are recommended by BASHH and are able to narrow the testing window period to 4 weeks compared to the older 3rd generation tests where the window period is 12 weeks. They also have the added advantage that they require a smaller (50 µl) venous blood sample.
In addition, the 4th generation tests are able to detect the p24 antigen, which is a core protein in the HIV virus and is detectable before the first occurrence of HIV-specific antibodies and its presence is representative of a recent seroconversion. Although the 3rd generation tests are less sensitive than the 4th generation tests, some are available that utilise saliva samples, offering a convenient alternative to blood sampling.
When interpreting HIV diagnostic tests, consideration should be given to the fact that HIV testing immediately after a possible transmission will not detect HIV antibodies. Use of HIV RNA viral load testing, may detect earlier HIV infection [up to 10 days earlier] in patients who have symptomatic “seroconversion illness”. Specialist advise should be sought to advise on the HIV testing in this scenario.
HIV infection cannot be fully excluded until at least 4 weeks after a possible HIV exposure using a 4th generation test and after 4 weeks after completing post exposure prophylaxis [PEPSE].
National Guidelines for testing are from 2008 so are due for update. BHIVA

www.avert.org: Accessed June 2019
Global situation
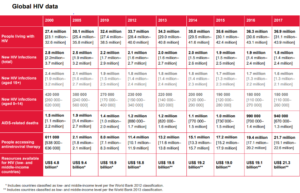
There were approximately 37 million people in the world living with HIV at the end of 2017 and of these, 9.4 million do not know they have the virus and need to be reached with HIV testing services, and around 15 million do not have access to HIV treatment, including 1.8 million children.
Since the start of the epidemic, around 77 million people have become infected with HIV and 35 million people have died of AIDS-related illnesses.
Sub-Saharan Africa is the most affected region, with 25.8 million people living with HIV in 2017 and accounts for almost 65% of the global total of new HIV infections and nearly 1 in 20 adults is affected.
But it is not all doom and gloom, the world has exceeded the AIDS targets of Millennium Development Goal 6 (MDG), halting and reversing the spread of HIV, which were set in 2000 and achieved in 2015. The global response to the MDG 6 has averted 30 million new HIV infections and nearly 8 million AIDS-related deaths since the MDGs were set. More and more countries are getting on the Fast-Track to end the AIDS epidemic by 2030 as part of the Sustainable Development Goals (SDGs). The epidemic has been forced into decline.
Global situation
New HIV infections have been reduced by 47% since the peak in 1996. In 2017, around 1.8 million were newly infected with HIV, compared to 3.4 million in 1996. Since 2010, new HIV infections among adults have declined by an estimated 16%, and among children by 35%.
AIDS-related deaths have been reduced by more than 51% since the peak in 2004. In 2017, around 940 000 people died from AIDS-related illnesses worldwide, compared to 1.9 million in 2004 and 1.4 million in 2010.
Ensuring access to antiretroviral therapy for nearly 22 million people is an achievement deemed impossible 18 years ago. In 2000, fewer than 1% of people living with HIV in low- and middle-income countries had access to treatment, in 2010 only 8 million people had access to treatment. In 2017, the global coverage for receiving antiretroviral therapy was 59% in adults, 52% in children and 80% in pregnant women.
UNAIDS has developed a Fast-Track approach called 90-90-90, to reach a set of time-bound targets by 2020.5 The targets include:
90% of all people living with HIV knowing their HIV status
90% of people who know their HIV-positive status having access to treatment
90% of people on treatment having suppressed viral loads
Global situation
They also include reducing new HIV infections by 75% and achieving zero discrimination.In 2017, the global results were:
75% of all people living with HIV knew their status
79% of those were accessing treatment
81% of those on treatment were virally suppressed
At time of writing 7 countries including the UK have already achieved the 90-90-90 targets ahead of 2020 schedule with many more on track. They are Denmark, Botswana, Cambodia, UK, Eswatini, Namibia and the Netherlands,
But HIV continues to shine a harsh light on the inequalities of the world. AIDS is unfinished business, the number of people living with HIV continues to increase, in large part because more people globally are accessing antiretroviral therapy and as a result are living longer, healthier lives but there is still an unacceptably high number of new HIV infections (especially in young girls age 15-19 in Sub Saharan Africa where they account for three out of four new infections) and AIDS-related deaths occurring each year. Stigma against HIV people is still at an unacceptably high level and is one of the most difficult aspects of the disease for those living with HIV.
UNAIDS factsheets 2017. Accessed June 2019 6
HIV in the UK
The latest data from 2018 is very positive.
The UK celebrated being one of only 7 countries in the world to have met the UNAIDS 90-90-90 targets ahead of the 2020 completion date.
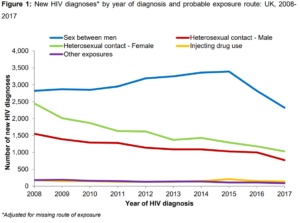
In addition, there had been a substantial decline in the rate of new HIV diagnoses
This recent reduction has been mostly driven by fewer HIV diagnoses among gay and bisexual men, which have decreased by almost a third since 2015.
HIV in the UK
Up until 2015 rates had been increasing year on year so this has been a monumental shift in the epidemic. It is most likely driven by a number of factors:
Namely by a large increase in HIV testing particularly in at high risk gay and bisexual men
Improved access and uptake of ARVs.
Use of PrEP has had an additional effect but as it is a relatively new intervention its true effect has still to be fully measured.
There have also been steady reductions in new diagnoses among black African and black Caribbean heterosexuals over the past decade from 2,655 in 2008 to 594 in 2017. This is largely due to changes in migration patterns, with fewer people entering the UK from high prevalence countries.
For the first time, there has also been a drop in the number of diagnoses reported among other heterosexuals, which fell by 20% in 2017. Previously, diagnoses had remained stable at around 1,000 diagnoses per year in this group. The cause of this change is uncertain at present. Continued low numbers of HIV diagnoses were reported in injecting drug users (140) and people who acquired HIV through other exposure routes (90), such as mother to child transmission and recipients of blood/tissue product.
HIV in the UK
London continues to have the highest rates of HIV in the country with 36% of all new diagnoses and 39% of people accessing HIV care in 2017. Lambeth is the London borough with the highest proportion of residents who have been diagnosed with HIV at 15 per 1,000.
The overall mortality rate for people aged 15-59 who were diagnosed early was, for the first time, equal to that of the general population for the same age group
Despite this good news, significant challenges remain to the control of HIV in the UK. The proportion of people diagnosed at a late stage of infection (CD4 count< 350cells/mm3) has remained persistently high over the past five years at close to 40%. Late diagnosis is associated with a ten-fold increased risk of short-term mortality (within a year of diagnosis) and an increased risk of onward transmission, with people diagnosed late having been unaware of their HIV infection for approximately three to five years.
The overall late diagnosis rate in 2017 was 43% (1,352/3,118), with the highest rates among black African heterosexual men and the lowest rates among gay and bisexual men (33%; 530/1,586). In 2017, there were 230 people with an AIDS-defining illness reported at HIV diagnosis and 428 deaths among people with HIV.
HIV in the UK
The population of people living with diagnosed HIV infection receiving care (93,385) is growing older and diversifying. In 2017:
39% of people were aged 50 years or above.
14% of gay and bisexual men receiving care were from black, Asian and other minority ethnic (BAME) groups
26% of heterosexuals receiving care were white.
These figures were 18%, 12% and 21%, respectively, in 2008.
Of the 4,363 people diagnosed in 2017, 53% were gay or bisexual men and of the 1,810 heterosexual people, 38% were black African men and women.
The ageing cohort of people living with HIV emphasises the importance of integrated care pathways to manage co-morbidities and other complications such as polypharmacy and the consequential potential drug interactions.
Data from Public Health England. Accessed June 2019
Click here for part 3